LiFePO4 vs. Lead-Acid: How Large is the Discharge Speed Gap?
Battery Discharge Response Time
| Battery Type | Internal Resistance | 0–200A Response Time | Characteristics |
|---|---|---|---|
| LiFePO4 (Lithium Iron Phosphate) | Low | < 10ms | Instantaneous material response |
| Lead-Acid (AGM/EFB) | High | 100–500ms | Slower chemical reaction |
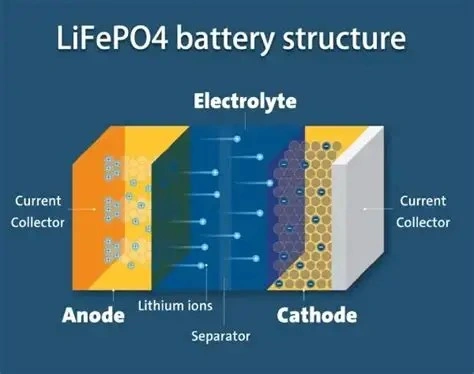
Many drivers report that their car feels more responsive—specifically during initial acceleration—after upgrading to a lithium battery. This is not a placebo effect. From the perspective of electrochemical kinetics, the speed at which a battery can release energy directly impacts the ignition coil’s saturation and the overall stability of the vehicle’s electrical system.
Why LiFePO4 is Faster
1. High Electrochemical Reversibility
The chemical reactions in a LiFePO4 battery are highly reversible with minimal side reactions. The lithium ions can intercalate (move into) and de-intercalate (move out of) the electrodes with very little resistance, allowing for near-instantaneous current delivery.
2. Electrolyte and Internal Resistance
Lithium batteries use high-conductivity organic solvents as electrolytes. Combined with the low-resistance design of the cell architecture, this allows for massive current flows without the significant voltage drops seen in lead-acid batteries.
3. Nano-scale Surface Area
The active material in LiFePO4 batteries is often engineered at a nano-scale level. This dramatically increases the surface area where the chemical reaction takes place, providing more “launch pads” for ions to move simultaneously.
The Limitations of Lead-Acid Chemistry
In contrast, lead-acid batteries rely on a slower, more cumbersome process:
- Lead Sulfate Deposition: As you draw power, lead sulfate (PbSO4) forms on the electrode plates. This layer acts as an insulator, progressively slowing down the reaction.
- Limited Surface Area: The active particles are much larger than those in lithium batteries, meaning fewer ions can react at any given millisecond.
- Electrolyte Resistance: Diluted sulfuric acid is a poorer conductor than the organic salts used in lithium batteries, leading to the higher internal resistance that characterizes the 100-500ms lag.
Summary
For modern vehicles with complex electronic fuel injection (EFI) and high-energy ignition systems, a faster battery means the spark plugs receive a more consistent and powerful surge of energy at the exact moment they need it. This results in more complete combustion, crisper throttle response, and a more efficient engine.